How Lithium Polymer Batteries are Made
Great Power Batteries
As part of the 2014 China trip, I got the chance to tour one of our battery manufactures. Robert was kind enough to give me a tour of Great Power Battery and to hook me up with one of the material engineers that works there. The following is a rough step-by-step of how LiPo batteries are manufactured.
Great power has a multi-building campus outside of Zhuhai, China.
Robert was nice enough to pick me up from the port of Zhuhai (pronounced chew-high) and give me a tour of the facility.
A quick step-by-step process of how LiPos are manufactured!
It was really neat to see the complexity of the uniforms and safety gear for the different positions along the assembly process.
Electrode Preparation
The material that will be coated on to the anode and cathode is mixed.
Large rolls of raw materials are loaded into the coating machine. I think this is aluminum.
A thin layer of the slurry of carbon and lithium material is applied in large square swaths onto the metal terminal material.
In a continuous fashion, the material is fed through an oven to cure the slurry onto the terminal. This room was probably 20m (65ft) long and pretty toasty.
There was two lines running continuously. One was the anode (I believe this is customarily aluminum) and one line was the cathode (copper). I remember this as a-a and c-c (copper is for cathode). Seen above is the never ending strip of copper with the carbon slurry cured in squares coming out of the oven. These squares will be cut and folded in the next few steps so don't be fooled into thinking they are making cells quite that big. Yet.
Lots of lots of raw electrodes ready for trimming.
This machine smashes the electrode like a pasta machine. In effect, making the sheets much thinner so that they can be folded more times.
Cutting the large sheets to proper width.
Electrode Creation
These machines take the rolls of electrodes, attach a terminal or tab, cut them to the proper length, and a whole bunch of other things I can't quite discern.
ReplaceMeOpen
ReplaceMeClose
Here's a video of the machines in glorious action.
And there's a ton of them.
ReplaceMeOpen
ReplaceMeClose
Here's a close up of the tab welding.
Once a pile of a given anode type is made, it is brought together with a pile of the cathode of the same size. A semi-permeable membrane is inserted between the layers, and the three strips are then folded together many times.
The above machine is a semi-automatic machine. It requires the operator to insert and line up the two electrodes, but it does the rest.
ReplaceMeOpen
ReplaceMeClose
Here's a video of the machine wrapping a cell.
ReplaceMeOpen
ReplaceMeClose
Above is a close-up of the folding or wrapping of the layers.
And when you're done you have the layers folded many times and taped.
Multiply the lines, and you can see how this factory produces 750,000 units a month.
ReplaceMeOpen
ReplaceMeClose
Above is a more automated wrapping machine. This level of automation makes me giddy. How do you build something like that and keep it running without constant tweaking?! Amazing.
Forming and Electrolyte
In a separate room, the housing is made.
These machines cut, form, and create the slightly flexible but protective housing for the inner electrodes and electrolyte polymer. I believe this material is a type of plastic or PVC.
The electrodes are then inserted between the protective housing and thermally welded together on three sides. The fourth side is left open for squirting in the electrolyte.
Electrodes in their housing.
In a carefully controlled environment the electrolyte is pumped into the cell. This part of the manufacturing floor is tightly controlled for humidity and temperature. The fume chamber is even more carefully controlled.
Multiple lines going at the same time. The final step of this line is thermally welding the fourth side of the battery to contain the electrolyte and seal the battery.
Charging
Once sealed, we can now call this thing a battery. This is the first charging cycle. You can see the plastic clips that clamp down on the exposed tabs. The small wing left on the battery is there to collect any electrolyte or gas that is pushed out during this first cycle.
Multiply by a lot of chargers.
And then fill an entire floor.
After their first charge is complete, the cells are strongly compressed. They told me "6 million pounds," but orders of magnitude often get mangled in translation. Let's call it a lot.
Air quickly degrades and causes problems with the battery chemistry. The heavy compression helps force out any remaining air from the cells.
I failed to get the exact amount of time or temperature, but the batteries bake compressed for a few hours.
The next step is to remove any excess electrolyte that has been forced out of the main cell and into the side pillow. If you've ever smelled a puffed or 'sweet' smelling battery, you know what this room smells like. Once the electrolyte is removed, the cell is trimmed.
At the end of all this, you have a battery. Technically, this is a single cell because the protection circuit and termination is still required.
Again, scale is important to remember. Every portable device we play with: tablets, cell phones, shavers, e-cigarettes, RC helicopers, etc. have a LiPo battery. Great power manufactures tens of thousands of cells a day.
Battery Creation
The protection circuit prevents the LiPo cell from being over charged (bad things happen when the cell is pushed past 4.3V), under charged (the cell can be harmed if pulled below about 2.8V), and short-circuit (shorting the positive and negative terminals together). If any of these triggers occur, the protection circuit very quickly disconnects the cell from the terminals.
Above, we finally have a formal battery with taped-in protection circuit (the yellow kapton tape) and positive and negative terminal wires (sans connector).
Dozens of different models of batteries were all over the factory. Above was a larger battery for a tablet.
I found this battery particularly intriguing. I've never seen a two part battery. Perhaps the dual cell allows for the battery pack to bend? Large cell goes under the keyboard and small cell behind the screen? I couldn't get more information about this one, but it does make me think about differently about battery options.
Each different battery model had their own line of employees.
Multiply by many lines.
Cylindrical Coverings
On a different line Lithium Iron Phosphate batteries (LiFePO4) were being made. I was told the process is nearly the same, so I didn't get as many photos. Because "lithium iron phosphate cells are much harder to ignite in the event of mishandling" they are considered the 'safer' battery compared to LiPo batteries. I'm not entirely convinced, but LiFePO4 batteries are finding their way into many rechargeable vehicle applications. Watch out for the nominal 3.2V (instead of 3.7V for LiPos), but if you're making a massive battery pack of cells in series, 3.2V doesn't really matter.
ReplaceMeOpen
ReplaceMeClose
This machine would take bare cells and automatically cut and slip on the heat shrink label. The loose cells then entered a 4 foot heat tunnel. I just love the sound in this video.
After exiting the oven, a AA sized battery complete with heat shrink label.
Battery Testing
After asking so many questions and taking so many photos the materials engineering was really getting into showing me around. I don't think I was much like their average tour. Excitedly, the engineer asked me if I would like to see the test chamber - YES!
On a different floor of the building a caged room housed eight machines of medieval torture. QC and failure testing is a part of every batch of batteries produced during the day.
They quickly offered me access and I hesitantly entered. Above is the oven that pushes batteries to their eventual demise.
The aptly named 'battery crush tester' takes a cell and squeezes the sides laterally until it fails.
The puncture tester is composed of a 10kg mass with a 10mm diameter rod at the end of it. The test pulls this mass 1m into the air and thrusts it down into the flat face of the cell. The engineer fired this one with an astounding thud that caught me off guard. It's a lot of force.
The stake test. Can we poke it until it ignites?
The fold test. This presses the cell down onto a length of triangular steel that causes the cell to fold at gnarly angles. Needless to say the carnage was epic and the chambers were covered in the soot of failed cells.
A few cells after their voyage.
Once they reach the end of the test, the cells are decomposed, unrolled, and the failure vector evaluated. Overall I was impressed with the level of undertaking and verification.
Big Batteries
The final stop on the tour was is a room far from the others, on the opposite end of the building.
This is where the very large battery packs are built and tested. I was told these are used to power an electric bus.
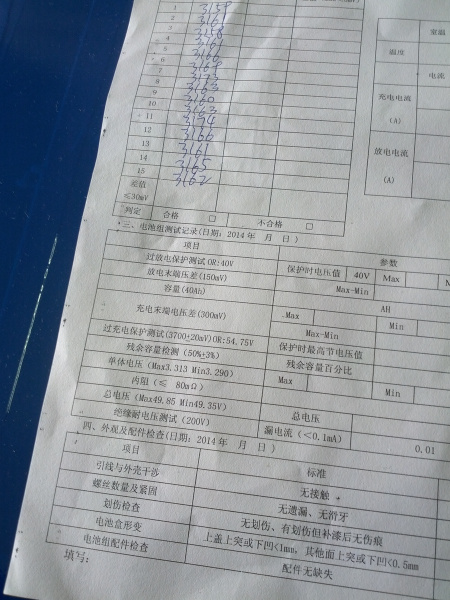
Imagine what you could do with 49V at 40,000mAh. The largest packs were 100Ah. That's nearly 5000Wh! Incredible stuff.
Resources and Going Further
We hope you've enjoyed reading! These tutorials take a lot of work and we hope you learn something from it! If you liked reading please leave a comment and let us know.
Ready to play with some batteries? Here are a few of our products to checkout:
- Polymer Lithium Ion Battery - 2000mAh
- Polymer Lithium Ion Battery - 400mAh
- USB LiPoly Charger - Single Cell
- LiPo Charger Basic - Micro-USB
- "Uh-oh" Battery Level Indicator Kit
Now that you've read how lithium based batteries are made, here are some tutorials that may strike your fancy: